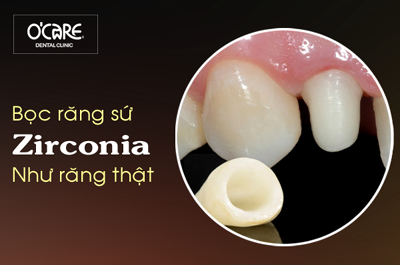
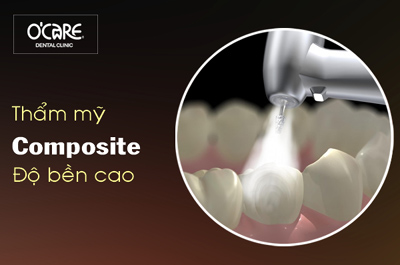
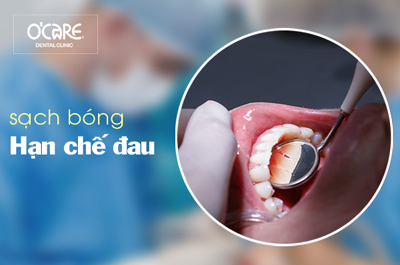
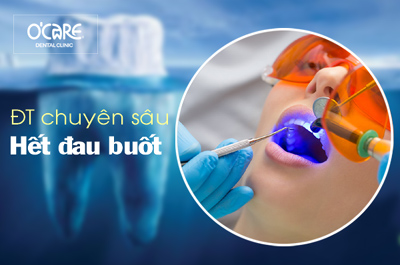
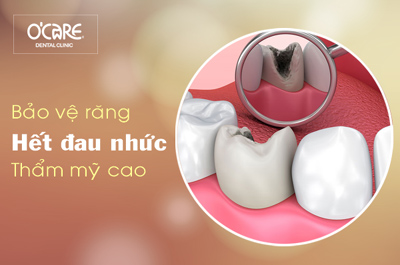
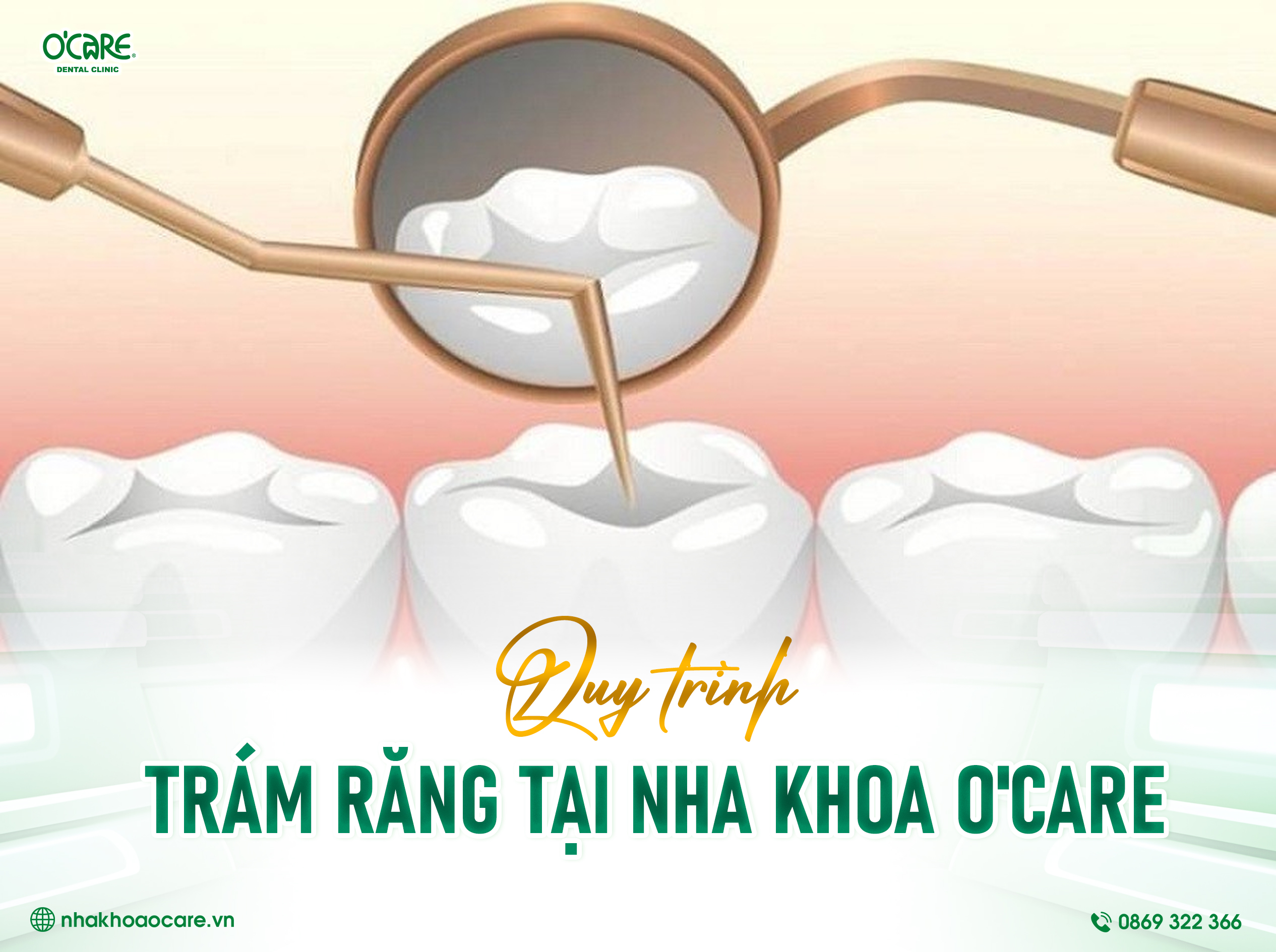
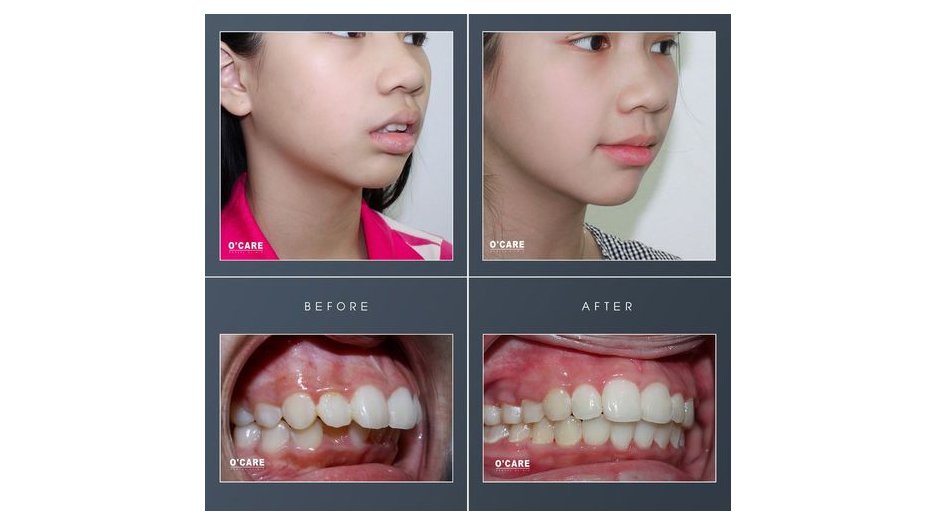
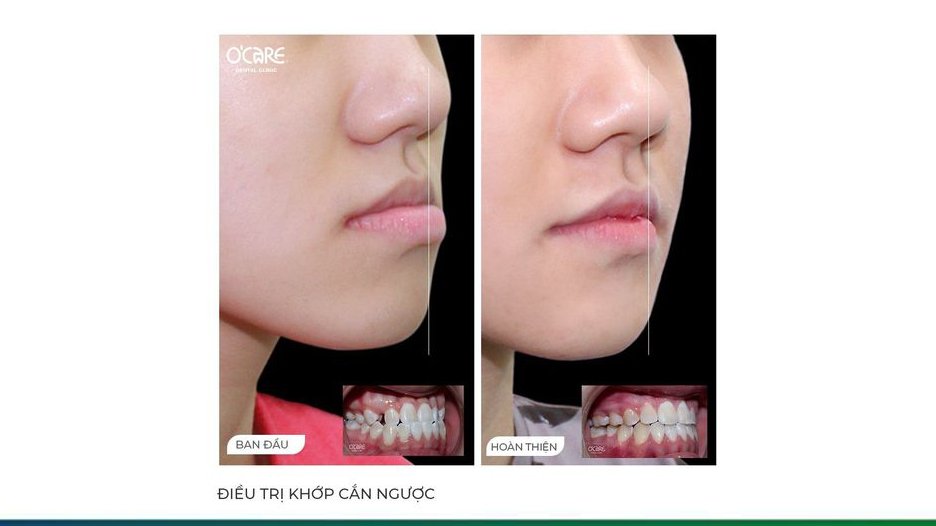
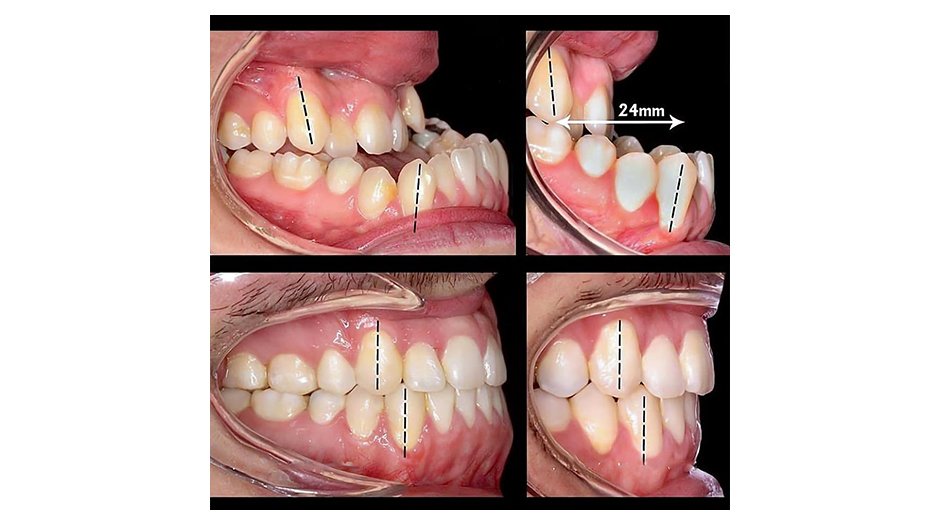
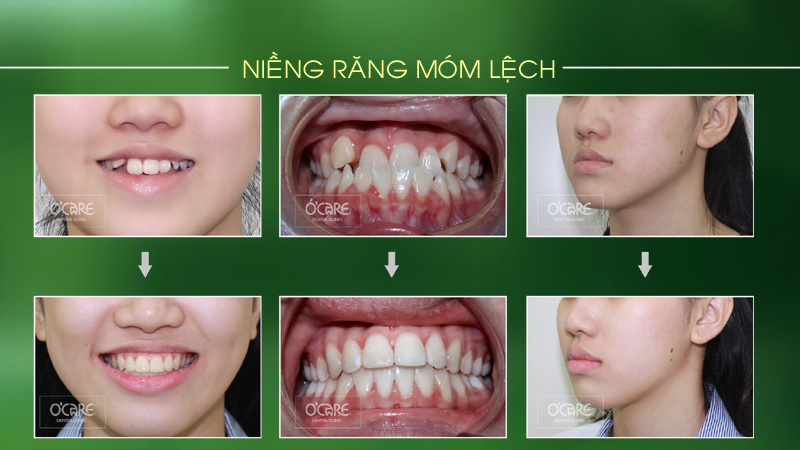
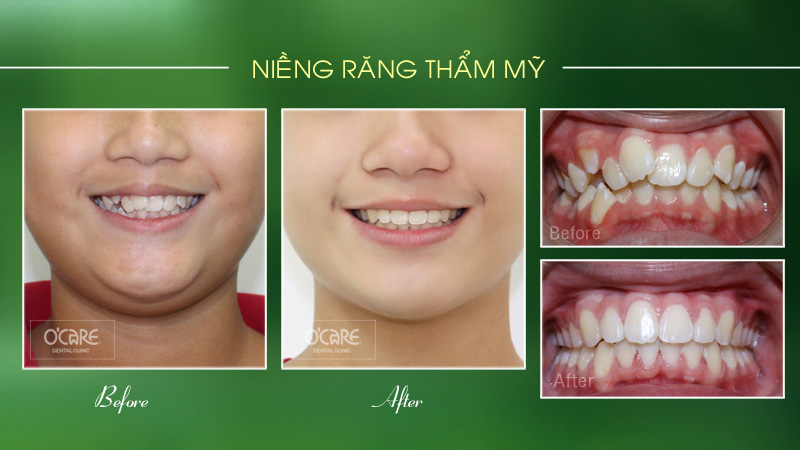
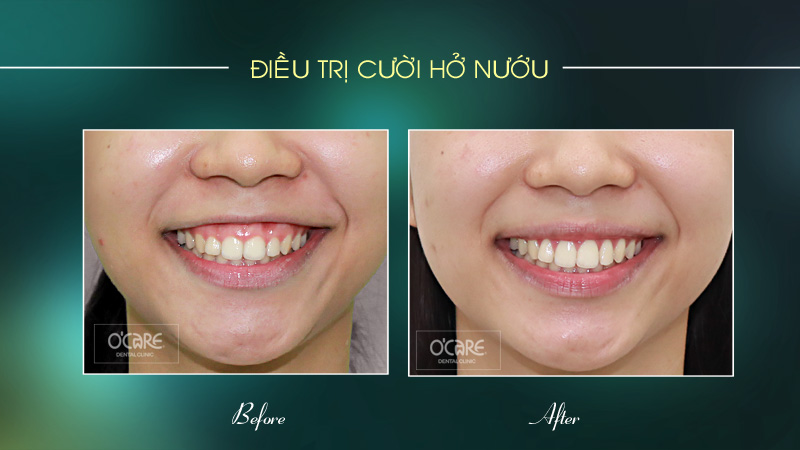
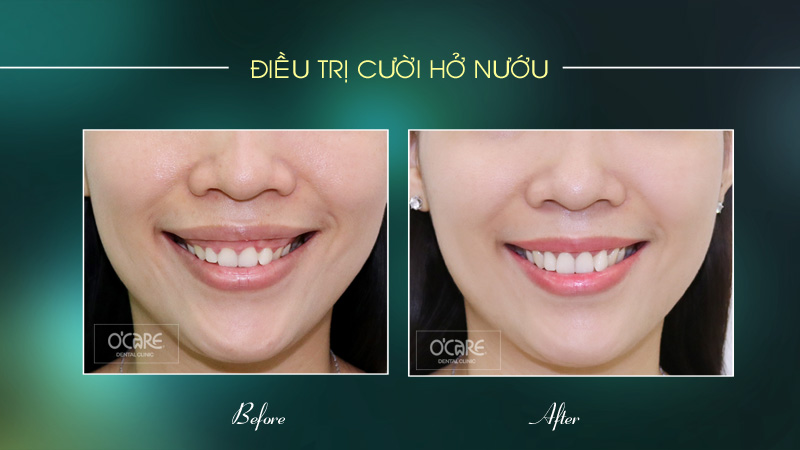
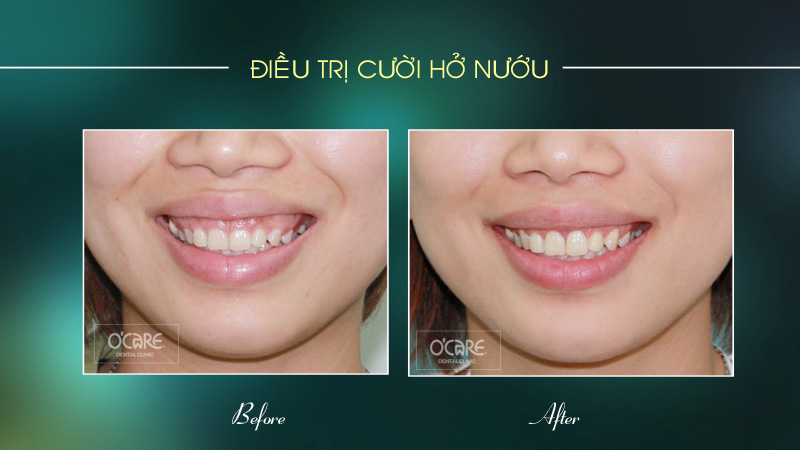
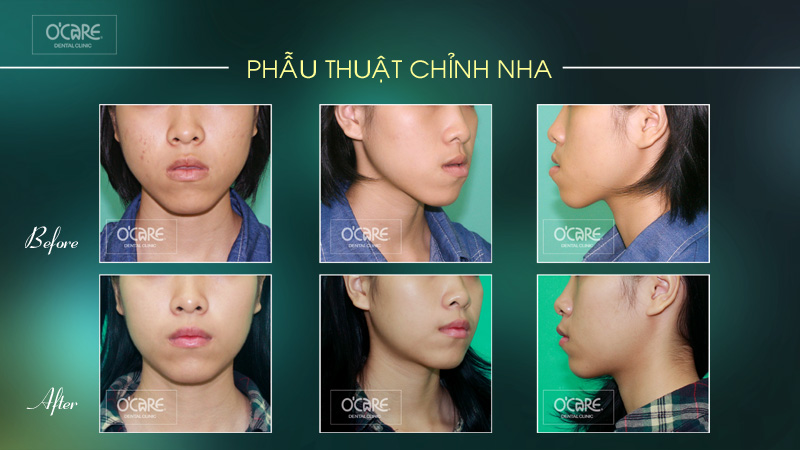
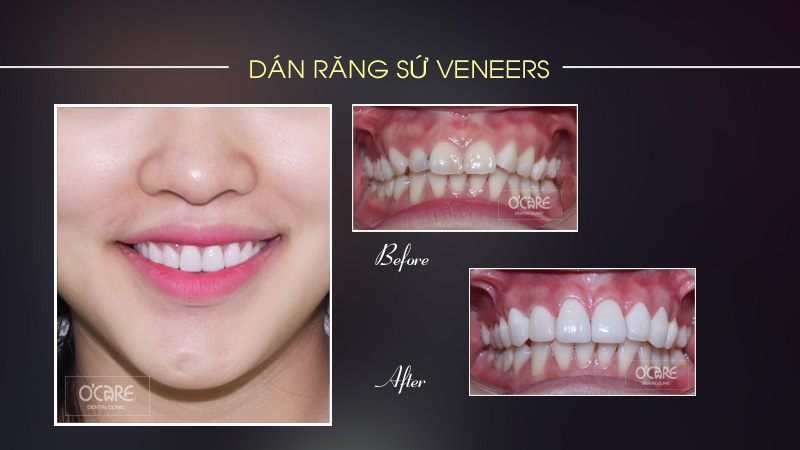
Tin tức & Sự kiện
-
 Nha khoa O’Care thông báo lịch làm việc & nghỉ lễ Giỗ Tổ Hùng Vương
Nha khoa O’Care thông báo lịch làm việc & nghỉ lễ Giỗ Tổ Hùng Vương
-
 Nha khoa O’Care Hà Nội đồng hành cùng sinh viên trường Đại học Lao động – Xã hội trên hành trình chăm sóc sức khỏe và thẩm mỹ nụ cười
Nha khoa O’Care Hà Nội đồng hành cùng sinh viên trường Đại học Lao động – Xã hội trên hành trình chăm sóc sức khỏe và thẩm mỹ nụ cười
-
 Nha khoa O’Care tham dự lễ kỷ niệm 93 năm thành lập Đoàn TNCS Hồ Chí Minh tại trường Đại học Lao động – Xã hội
Nha khoa O’Care tham dự lễ kỷ niệm 93 năm thành lập Đoàn TNCS Hồ Chí Minh tại trường Đại học Lao động – Xã hội
-
 [RECAP] Sự kiện “Thăm khám răng miệng và Lấy cao răng miễn phí” hưởng ứng Ngày sức khỏe răng miệng Thế giới của nha khoa O’Care Hà Nội
[RECAP] Sự kiện “Thăm khám răng miệng và Lấy cao răng miễn phí” hưởng ứng Ngày sức khỏe răng miệng Thế giới của nha khoa O’Care Hà Nội